- Blog
- Drake childs play without intro
- Obd2 chip tuning box
- Leica geosystems
- In christ alone lyrics i place my trust
- Toad diagnostic software frostwire
- Cancion del mariachi tribal
- Cinematic strings 2 versus cs 2 review
- National lampoon van wilder rise of taj
- Diddy press play songs
- How to make background transparent in coreldraw x5
- Hindi movie saudagar 1973 songs
- Dbz all episodes english
- How to clean puch moped body
- Rabaran rudki gujarati song
Maternal morbidity of VBAC-2 was comparable to RCS. Comparing VBAC-2 versus RCS, the hysterectomy rates were 0.40% versus 0.63% (P = 0.63), transfusion 1.68% versus 1.67% (P = 0.86) and febrile morbidity 6.03% versus 6.39%, respectively (P = 0.27). VBAC-2 success rate was 71.1%, uterine rupture rate 1.36%, hysterectomy rate 0.55%, blood transfusion 2.01%, neonatal unit admission rate 7.78% and perinatal asphyxial injury/death 0.09%. Meta-analyses were performed using RevMan-5 to compare VBAC-1 versus VBAC-2 and VBAC-2 versus RCS. Two independent reviewers selected studies and abstracted and tabulated data and pooled estimates were obtained on success rate, uterine rupture and other adverse maternal and fetal outcomes. No randomised studies were available, case series or cohort studies were assessed for quality (STROBE), 20/23 available studies included.


We searched MEDLINE, EMBASE, CINAHL, Cochrane Library, Current Controlled Trials, HMIC Database, Grey Literature Databases (SIGLE, Biomed Central), using search terms Caesarean section, caesarian, C*rean, C*rian, and MeSH headings 'Vaginal birth after caesarean section', combined with second search string two, twice, second, multiple. To perform a systematic review of literature on success rate of vaginal birth after two caesarean sections (VBAC-2) and associated adverse maternal and fetal outcomes and compare with commonly accepted VBAC-1 and the alternative option of repeat third CS (RCS).
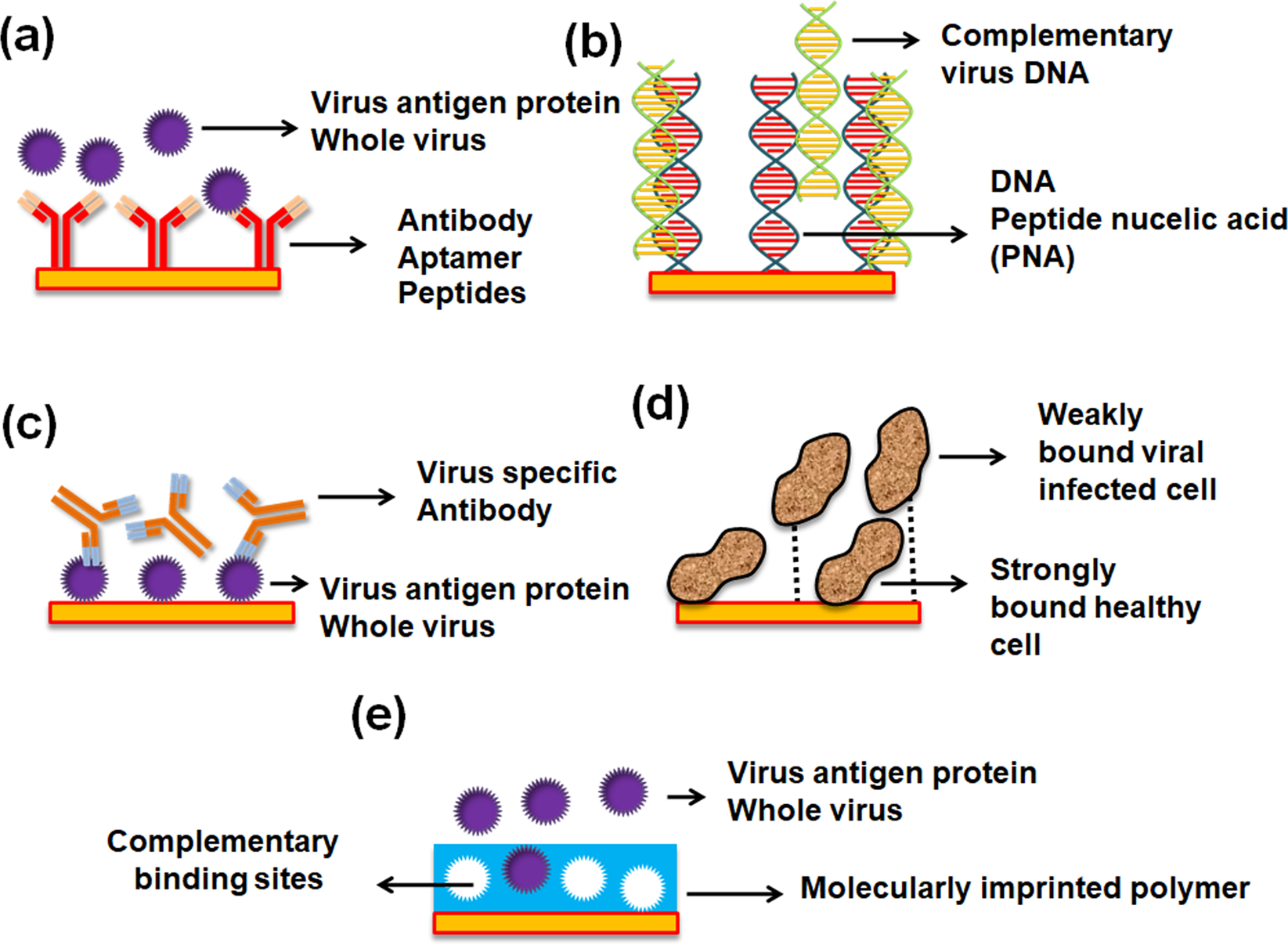
#Cinematic strings 2 versus cs 2 review trial#
Trial of vaginal birth after Caesarean (VBAC) is considered acceptable after one caesarean section (CS), however, women wishing to have trial after two CS are generally not allowed or counselled appropriately of efficacy and complications.
- Blog
- Drake childs play without intro
- Obd2 chip tuning box
- Leica geosystems
- In christ alone lyrics i place my trust
- Toad diagnostic software frostwire
- Cancion del mariachi tribal
- Cinematic strings 2 versus cs 2 review
- National lampoon van wilder rise of taj
- Diddy press play songs
- How to make background transparent in coreldraw x5
- Hindi movie saudagar 1973 songs
- Dbz all episodes english
- How to clean puch moped body
- Rabaran rudki gujarati song
